Integrated Delivery Networks: Direct Contracting and Medication Management
Highlights of the report:
Download a PDF of these Highlights
Integrated deliver networks' (IDN) contracting and medication management initiatives in 2026 are increasingly shaped by where they have the greatest influence across the care continuum. HIRC's report, Integrated Delivery Networks: Direct Contracting and Medication Management, reviews the landscape of direct contracting between IDNs and pharmaceutical manufacturers and examines health systems' control over physician prescribing behavior. The report addresses the following:
- What are the recent trends in IDNs' acquisition of pharmaceuticals?
- In what cases will IDNs seek to purchase medications directly from manufacturers rather than their GPO?
- What is the landscape of direct purchasing contracts between IDNs and manufacturers? Which therapeutic classes have the most direct contracting activity?
- What types of direct contracts are most common and what are the range of discount/ rebate amounts? Which manufacturers are most willing to offer direct contracts?
- What is the status of IDN pharmacy services, including the prevalence of IDN-owned specialty pharmacies?
- What medication management tactics do IDNs utilize and how are they enforced?
- What is the status of biosimilars adoption among IDNs?
Key Finding: IDNs’ contracting and medication management approaches in 2026 are shaped by a complex operating environment, with direct contracting used in targeted scenarios and formulary strategies adapting to payer-driven access requirements.
Direct Contracts Between Manufacturers and IDNs Most Common for Vaccine Products. While IDNs continue to source most of their pharmaceutical product from their affiliated group purchasing organizations (GPOs) and wholesalers, IDNs also purchase directly from manufacturers situationally, especially during shortages or when there is a clear financial advantage. In 2026, IDNs most often report direct contracts in annual vaccines (e.g., flu, COVID, pneumococcal), followed by diabetes, oral breast cancer, and immunoglobulins.
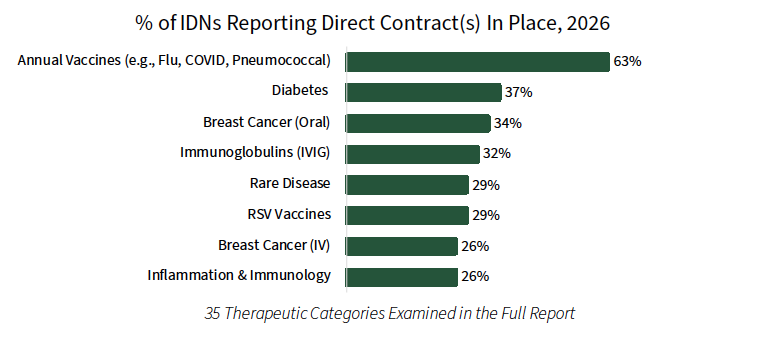
The complete report reviews the direct contracting landscape (prevalence, contract types/approaches, and discount amounts) for a listing of 30+ therapeutic areas.
A Majority of IDNs Have an Inpatient Formulary and Stable Adoption of Biosimilars. IDNs use a number of levers and tools to manage medication costs and influence physician prescribing behavior. Most IDNs have an inpatient formulary (86%) and standardized treatment pathways (61%), and 46% report having an outpatient formulary. Biosimilars are now the standard of care across most IDNs, though adoption has stabilized as IDNs balance payer formularies, physician preferences, operational complexities, and reimbursement dynamics.
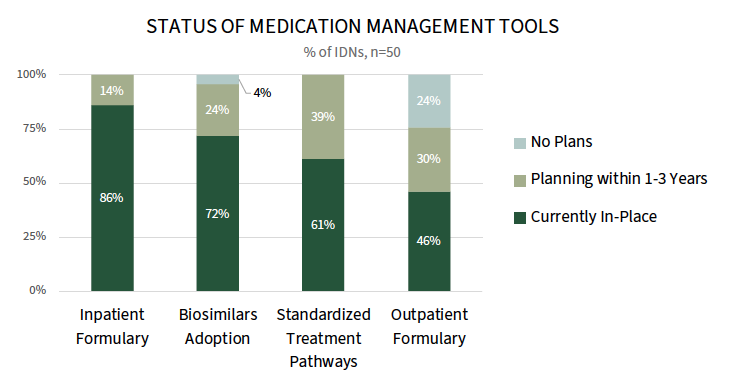
To enforce the use of formularies and pathways, IDNs embed order sets and best practice alerts into the electronic medical record. Some IDNs will also monitor prescribing patterns and follow-up with provider education to encourage compliance.
The full report examines the following:
- Status of preferred inpatient products across 25+ traditional, specialty, and oncology therapeutic categories
- Status of preferred outpatient products across 25+ traditional, specialty, and oncology therapeutic categories
- Status of biosimilars adoption across 15+ reference brands
Research Methodology and Report Availability. In December and January, HIRC surveyed 50 IDN pharmacy directors, medical directors, and senior leaders. Online surveys and follow-up telephone interviews were used to gather information. The full report, Integrated Delivery Networks: Direct Contracting and Medication Management, is part of the Organized Providers Service, and is now available to subscribers at www.hirc.com.
Download a PDF of these Highlights
Download Full Report (Subscribers only) >


