Rare Disease & Gene Therapy: Commercial MCO Management, Financing, & Mfr. Engagement
Highlights of the report:
Download a PDF of these Highlights
A growing volume of innovative rare disease and gene therapy treatments continue to enter the market, requiring novel utilization management, financing, and contracting approaches. HIRC’s report, Rare Disease & Gene Therapy: Commercial Health Plan Management, Financing, and Manufacturer Engagement, examines plans' handling of high and ultra high-cost therapies across 20 rare disease states and evaluates best-in-class manufacturer support. The report addresses the following questions:
- How are plans working to manage the cost and utilization of rare disease and gene therapies in 2025/2026?
- What specific utilization and formulary management tactics are applied across 20 unique rare disease categories?
- Which financial protection programs are health plans and their self-funded employer clients exploring for rare disease medications?
- What is the nature of the contracting environment across 20 broad rare disease states? How interested are commercial MCOs in novel contracting arrangements for rare disease & gene therapies?
- How are manufacturers supporting commercial plans and patients in rare disease?
Key Finding: Commercial MCOs continue to manage access to rare disease medications primarily through stringent prior authorization criteria; however, in therapeutic classes with multiple available products, plans are increasingly able to implement preferred drug strategies and step therapy.
Prior Authorization Criteria and Value-based Contracting Top Commercial MCOs' Efforts to Manage Rare Disease Spend. Commercial MCOs are currently managing the cost and utilization of rare disease and gene therapies primarily through robust PA clinical requirements that often consider the clinical trial inclusion/exclusion criteria. Plans are also focused on assessing and pursuing financial and contracting solutions, such as outcomes-based contracts, traditional access-based contracts where possible, as well as financial protection program solutions. Provider narrow network and reimbursement strategies also play a large role in plans' management of rare disease and gene therapy medications.

The full report examines plans' tactics in detail across 20 rare disease categories.
Financing & Contracting Considerations in Rare Disease. Commercial MCOs are evaluating a variety unique financial protection programs, benefit designs, and other methods to finance rare disease & gene therapies, discussed in the complete report. When it comes to contracting with manufacturers, approaches vary by therapy type. Traditional access-based contracts are most often reported in pulmonary arterial hypertension (PAH) and hemophilia, while a higher prevalence of risk/outcomes-based contracts are observed for gene therapies.
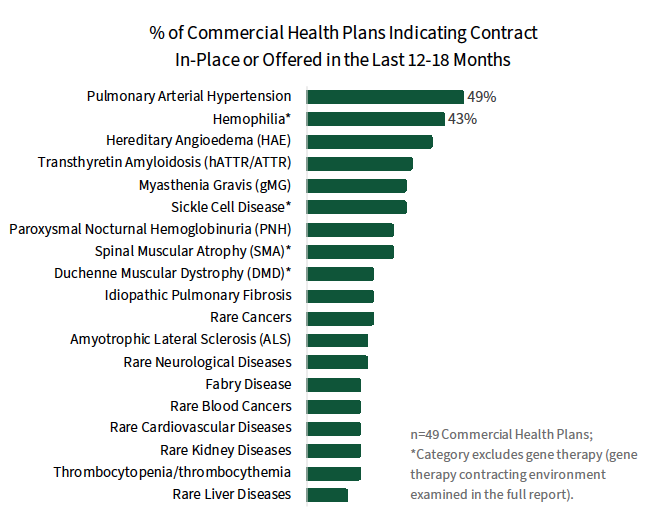
The full report examines the rare disease contracting environment in detail, as well as plans' interest in prevalence-based rebates, value/outcomes-based contracts, and pay-over-time arrangements.
Novartis, Genentech, and Pfizer are Among Leaders in Rare Disease Engagement with Commercial MCOs. Plans were asked to consider and provide a best-in-class manufacturer nomination across a number of rare disease engagement parameters, including those noted below. Novartis, Genentech, and Pfizer are among those nominated as best across categories.
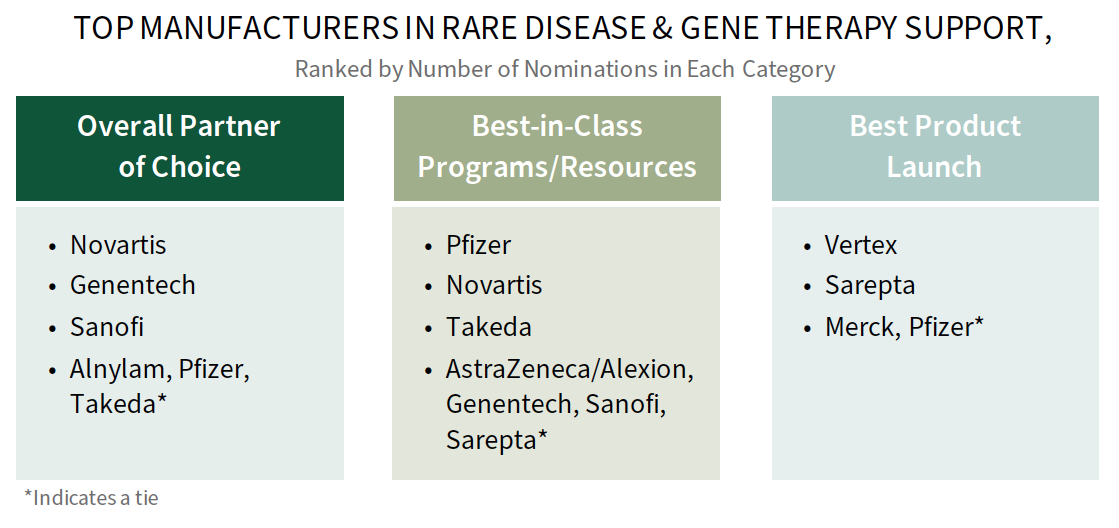
Many of the leading manufacturers in rare disease engagement have growing or robust rare disease portfolios, where products are accompanied by significant support services and novel contracting arrangements. The complete report provides the full listing of manufacturers recognized, drivers of best-in-class nominations, and a commercial MCO needs assessment.
Research Methodology and Report Availability. In July/August, HIRC surveyed 49 pharmacy and medical directors from national, regional, and BCBS plans representing 110 million lives. Online surveys and follow-up telephone interviews were used to gather information. The Rare Disease & Gene Therapy: Commercial Health Plan Management, Financing, and Manufacturer Engagement report is part of HIRC's Special Report Series, and is now available to subscribers at www.hirc.com.
Download a PDF of these Highlights
Download Full Report (Subscribers only) >


