Pharmacy Benefit Managers: Oncology Medication Distribution, Formularies and Contracting
Highlights of the report:
Download a PDF of these Highlights
Pharmacy benefit managers (PBMs) are tightening utilization management and formulary strategies to more effectively manage oncology spend in 2026 and beyond. HIRC's report, Pharmacy Benefit Managers: Oncology Medication Distribution, Formularies, and Contracting, examines PBMs' oncology medication management approaches and the resulting contracting environment across oncology therapeutic areas. The report addresses the following questions:
- What are the recent trends and level of PBM involvement in oncology drug dispensing and distribution?
- Which oncology medication management tactics are utilized by PBMs?
- What is the status of and future expectations for oncology formularies and excluded product lists?
- What is the perceived contracting environment across oncology medication types?
- What is the estimated level of rebates firms offer to compete for formulary access?
- Which manufacturers are most often nominated as PBMs' Partner of Choice? Which are the most willing to contract?
Key Finding: Over the next 12-18 months, PBMs are looking to enhance use of UM tools such as tighter PA requirements, step therapy, limited first fills, and quantity limits to better manage oncology drug costs for commercial and Medicare clients.
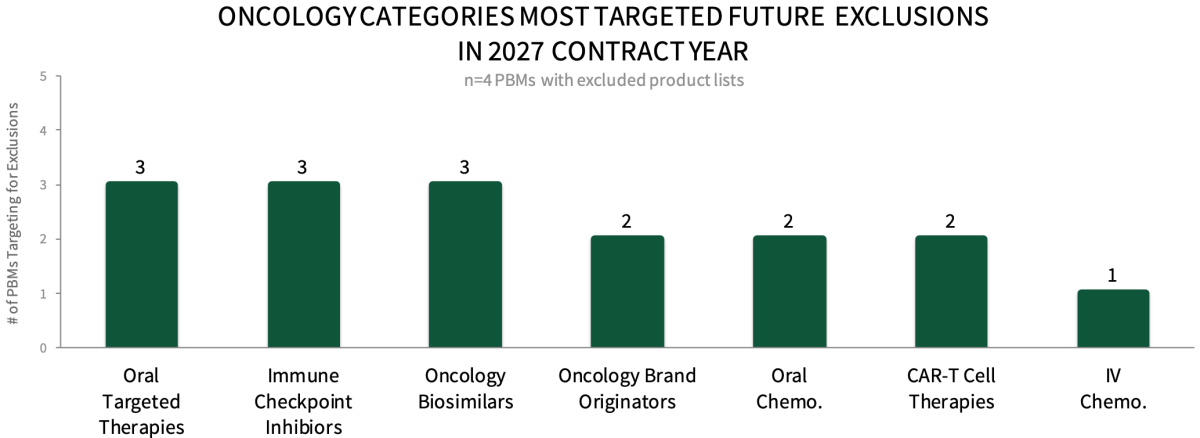
Oncology Medications Targeted for Additional Exclusions in 2027. Respondents were asked which oncology medication categories will be most targeted for future exclusions in the 2027 contract year. Panelists suggest that oral targeted therapies, immune checkpoint inhibitors, and oncology biosimilars will most often be targeted for future exclusions, followed by oncology brand originators, oral chemotherapies, and CAR-T cell therapies. The full report provides additional insights on PBM contracting expectations to avoid exclusion, as well as the factors driving formulary decision-making.
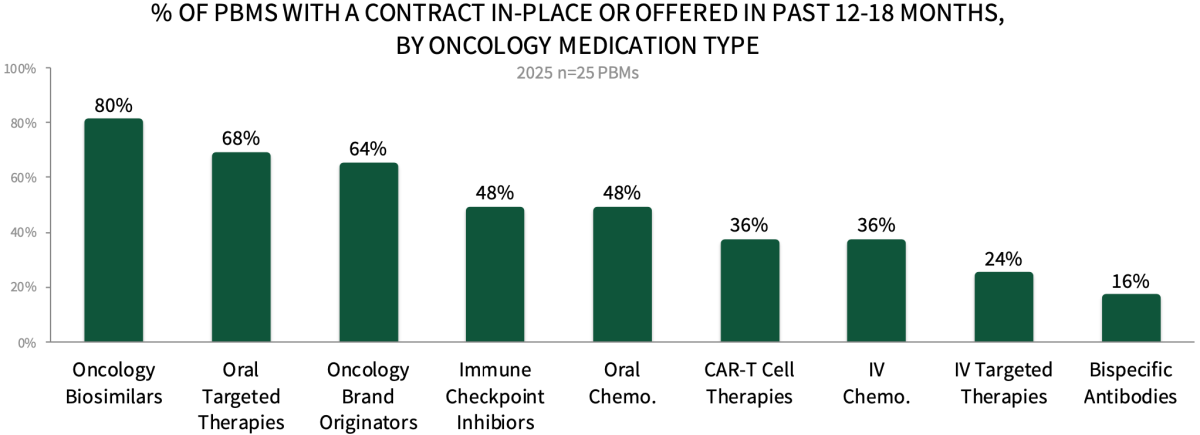
Contracting Environment for Oncology Medications. About 80% of PBM respondents in HIRC's sample report their organization currently has contract(s) in-place or has been offered a contract for oncology biosimilar medications. Additionally, over 45% of PBMs have a contract in-place or have been offered a contract for oral targeted therapies (68%), oncology brand originators (64%), immune checkpoint inhibitors (48%), and oral chemotherapies (48%). The full report examines contract approaches/types and most common discount/rebate amounts in each oncology category and across 19 unique cancer types.
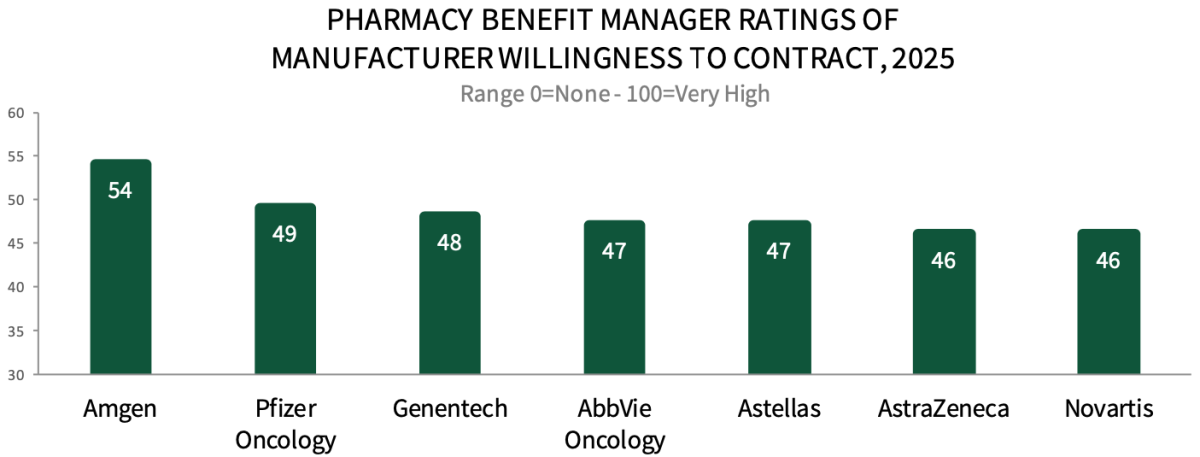
PBM Executives Rate Amgen As Most Willing to Contract in Oncology. Respondents were asked to consider a list of oncology medication manufacturers and rate their overall willingness to contract on a scale of 0=none to 100=very high. Amgen leads with the highest willingness to contract rating, followed by Pfizer Oncology, Genentech, AbbVie Oncology, Astellas, AstraZeneca, and Novartis. The full report provides a complete listing of PBM executives' ratings across a list of 40+ manufacturers, as well as nominations for partner of choice and best program/resource support in oncology.
Research Methodology and Report Availability. In September 2025, HIRC surveyed 25 pharmacy benefit manager key decision-makers. Online surveys and follow-up telephone interviews were used to gather information. The complete report, Pharmacy Benefit Managers: Oncology Medication Distribution, Formularies and Contracting, is available now to HIRC’s Managed Oncology subscribers at www.hirc.com.
Download a PDF of these Highlights
Download Full Report (Subscribers only) >

